Contents
ToggleCan Gluten Cause Gut Inflammation?
 Can gluten cause gut inflammation? It is well known that gluten causes celiac disease – an inflammatory condition of the small intestine. What is less well known, and often times completely ignored, is the connection between gluten and inflammation of other areas of the GI tract. Keep in mind that the GI tract is made up of the entire tube running from your mouth to your anus. Gluten induced inflammation has been medically researched and documented in the mouth, esophagus, stomach, small intestine, and large intestine. In other words, gluten can damage any area along the GI tract.
Can gluten cause gut inflammation? It is well known that gluten causes celiac disease – an inflammatory condition of the small intestine. What is less well known, and often times completely ignored, is the connection between gluten and inflammation of other areas of the GI tract. Keep in mind that the GI tract is made up of the entire tube running from your mouth to your anus. Gluten induced inflammation has been medically researched and documented in the mouth, esophagus, stomach, small intestine, and large intestine. In other words, gluten can damage any area along the GI tract.
Inflammatory bowel disease (IBD) is a broad term used to describe a variety of conditions that cause chronic inflammation in the digestive tract. Crohn’s disease and ulcerative colitis (UC) are commonly included under the IBD umbrella. But they’re not the only inflammatory bowel diseases. For example, celiac disease causes chronic gut inflammation. Others conditions include:
- Mouth Ulcers – AKA canker sores or aphthous ulcers
- GERD (chronic acid reflux)
- Esophagitis
- Diverticulitis
- Hemorrhoids
There’s no shortage of scientific evidence linking IBD to gluten. A recent systematic review and meta-analysis examined the results from 65 studies discussing the relationship between celiac disease and IBD (Crohn’s and UC specifically). It was determined that those with celiac disease are 9 times more likely to develop IBD than healthy controls. Suggesting that gluten-induced celiac disease may contribute to the development of IBD.
The meta-analysis also reported that those with IBD were almost 4 times more likely to develop celiac disease than healthy controls.
So the connection between celiac and IBD is strong. Strong enough that IBD patients should be screened for gluten sensitivity and vice versa. But unfortunately, this isn’t a popular idea in mainstream medicine.
Many doctors aren’t screening for gluten sensitivity. And only if “textbook” celiac disease is diagnosed do doctors recommend a gluten-free diet. In some cases, no diet changes are recommended at all. Instead, most IBD patients are given prescription medications that only mask symptoms and provide temporary relief.
So in this article, I’ll be discussing how gluten can trigger various inflammatory bowel diseases beyond celiac disease to highlight why a gluten-free diet should be considered more often. It might not solve every case, but I’ve witnessed it solve many.

How Gluten Contributes to Inflammatory Bowel Disease
Before I discuss specific conditions, I want to point out the key pathways in which gluten induces IBD.
Inflammation
As I mentioned above, gluten causes inflammation when consumed by individuals with gluten-sensitive genes. This sets off a cascade of events in the body, which include:
- Creating an overactive immune system
- Releasing inflammatory compounds that travel throughout the body
- Disrupting the balance of beneficial versus pathogenic gut bacteria
- Triggering your body’s stress response
As a result, all of the things outlined above can set the stage for autoimmune and inflammatory diseases throughout your body, including in your digestive tract.
Nutrient Deficiencies
Another way gluten contributes to IBD is through nutrient deficiencies. Gluten damages the lining of your intestines, which compromises digestion and nutrient absorption. Diarrhea is often a symptom of gluten sensitivity, which further prevents nutrients from being absorbed. Plus, most medications prescribed for IBD deplete your body of nutrients. And these nutrients play many critical roles within your digestive tract, which include fighting against inflammation as well as protecting and fueling the cells that line your intestines.
It’s worth noting that IBD further exacerbates your nutritional status. In fact, about 50% of people with IBD have nutrient deficiencies. Plus, medications used to treat IBD can cause nutrient deficits and make it more difficult to heal.
A few of the nutrients of concern and their connection to gut health are discussed below to give you a sense of how gluten-induced deficiencies can contribute to IBD.
- Zinc is a mineral that has hundreds of functions in the body. It’s a core component of many enzymes. It acts as an antioxidant to protect cells from free radical damage. And it’s involved in protein synthesis and immune function.
So zinc deficiency is linked to poor immune health. It’s also been associated with diarrhea and intestinal permeability in children with gastrointestinal diseases in developing countries.
- Vitamin A helps maintain the integrity of your intestinal cells as well as supports your immune system. Thus, vitamin A deficiency has also been strongly associated with diarrhea and intestinal permeability among children in developing countries.
- Vitamin D is essential for optimal immune health. It’s been shown to regulate the function of your gut bacteria as well as fight against inflammation. And this helps explain why vitamin D deficiency is associated with IBD.
- Magnesium is also a key player when it comes to immune regulation and inflammation. Plus, deficiencies have been shown to alter the composition of gut bacteria.
- Iron deficiency as well as excess iron in the gut due to malabsorption can lead to gut bacteria imbalances.
- Selenium has been shown to help regulate the inflammatory pathways involved in the progression of IBD. This suggests that a selenium deficiency could contribute to the development of disease.
- Glutamine is an amino acid that serves as a fuel source for the cells that line your digestive tract. It’s involved in the maintenance and repair of your gut lining to help maintain its integrity. Glutamine also protects your intestinal cells from damage and helps reduce gut inflammation. This explains why 30% of the glutamine your body makes or ingests is used by your intestines.
While your body can make glutamine from other amino acids, some people may not make enough due to poor health. For example, gluten-induced gut inflammation increases your body’s need for glutamine. This leads to what is referred to as conditional essentiality, and L-glutamine supplementation is often times necessary. Faulty digestion and malabsorption may also lead to a glutamine deficiency.
In addition, gluten-rich diets are filled with cereal for breakfast, sandwiches for lunch, and pasta for dinner. And these foods aren’t good sources of glutamine.
Additional nutrients of concern include vitamins B12 and K, folate, and calcium.

Gut Bacteria Imbalances
Foods with gluten are also high in calories and processed carbohydrates. Eating a diet rich in processed carbs leaves less room for more nutrient-rich foods. Weight gain and obesity are common manifestations of consuming processed gluten free foods.
These consequences can also cause gut bacteria imbalances that can trigger inflammation as well as reduce the microbial production of short-chain fatty acids, which feed the cells of your colon and help reduce gut inflammation.
For example, research has found that IBD patients have less bacterial diversity than controls as well as a smaller population of Bacteroides and Firmicutes, which are responsible for producing the short-chain fatty acid butyrate. Butyrate is the primary fuel source for the cells of the large intestine, and it has been shown to play important roles in maintaining the gut barrier, modulating inflammation, and aids in proper gut motility.
Effects of Glyphosate
Glyphosate, an herbicide heavily used on many grains including wheat and corn, has been associated with a possible increase in the incidence celiac disease (see diagram). And though correlation does not signify causation, this relationship should definitely be investigated further. I interviewed Dr. Stephanie Seneff on the potential side effects of this herbicide. You can listen to it here.
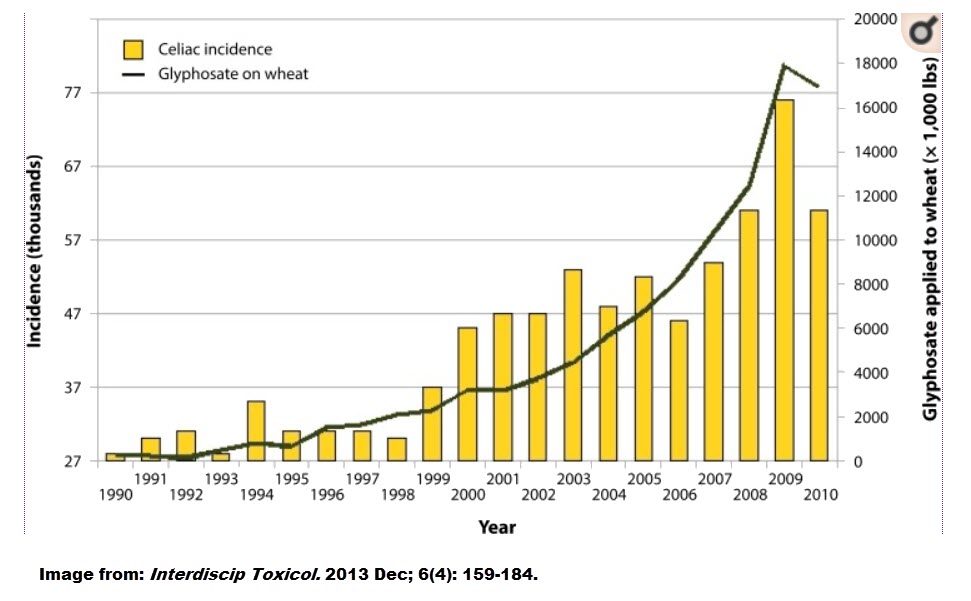
There’s also evidence that glyphosate, causes gut bacteria imbalances. It also binds to minerals (e.g., iron) and amino acids, which prevents them from getting absorbed and leads to nutrient deficiencies that can contribute to IBD.
In addition, research tells us that glyphosate significantly reduces the amount of manganese that plants absorb from the soil. Thus, eating a diet rich in processed foods is deficient in manganese. And this presents a problem because manganese is used by certain gut bacteria for a wide range of functions, which includes fighting against inflammation in the gut as well as cellular damage from free radicals.
Glyphosate may also be associated with a condition known as cholestasis, which reduces or restricts your body’s flow of bile, a condition common among patients with IBD.
Most recently, glyphosate has been linked to non-Hodgkin’s lymphoma, and cancer that can impact the digestive tract. The producer of glyphosate, Bayer, has been ordered to pay out 10 billion in damages over this connection.
Non Gluten “Gut Bombs” to Be Wary Of
Aside from gluten (and glyphosate), other herbicides/pesticides, processed foods and additives, and many medications can also contribute to a fundamental break down in gut function. Because most gluten free food options are highly processed, are not organic (contain herbicide and pesticide residue), and lead to gut damage that traditional doctors will typically try to medicate. It is best to try avoiding these items if you want to have optimal GI function.

Crohn’s Disease and Ulcerative Colitis
Crohn’s disease can impact any region of the digestive tract. But in most cases, the inflammation occurs within the small and large intestine.
Patients typically experience “flare-ups” followed by periods of remission. Some symptoms include:
- Abdominal pain
- Diarrhea (sometimes with blood in stool)
- Weight loss
- Anemia
- Gas and bloating
If untreated, the cycles of inflammation and scarring of intestinal tissue can ultimately obstruct the bowels as well as cause other severe complications.
Ulcerative colitis is the most common inflammatory bowel disease also characterized by “flare-ups” and periods of remission.
Unlike Crohn’s disease, UC specifically attacks the colon. The main symptom is bloody diarrhea, which can be accompanied by:
- Fatigue
- Weakness
- Weight loss
- Bowel urgency
- Abdominal pain
- Fever
The damage that occurs to the lining of the colon can cause waste and pathogens in the colon to be absorbed into the body and trigger an immune response, which causes more inflammation.
Contributing Factors to Crohn’s Disease and Ulcerative Colitis
Like celiac disease, Crohn’s and UC are caused by a dysfunctional immune system. Under normal conditions, your immune system attacks foreign invaders to protect your body from harm. But things go awry when your immune system starts attacking more than just bacteria and viruses.
For example, with celiac disease, your immune system misinterprets gluten. It sees it as a threat. So it attacks. And when it does, it causes inflammation in your gut and beyond.
So what causes the immune system to overreact when it comes to Crohn’s disease and UC? Like celiac, there’s usually a genetic component. But there’s also an environmental trigger. And gluten-induced inflammation, nutrient deficiencies, and gut bacterial imbalances are all possibilities.
What’s interesting is that Crohn’s disease and UC are more common in modern Western societies as well as in urban cities as opposed to rural areas. Possibly because people living in these regions are exposed to more environmental toxins and eat more heavily processed foods (that often contain gluten). They also adopt busy, stressful lifestyles with little time for exercise and relaxation.
In fact, studies have revealed that patients with Crohn’s disease eat more processed foods than those without. And evidence suggests doing so increases your risk of developing Crohn’s disease.
In this study, mice developed colitis within 2 days of being fed a high-sugar diet when compared to controls given a balanced diet. Plus, the mice had elevated levels of inflammatory markers, increased gut permeability, and decreased gut bacteria diversity.
This study found that 23% of Crohn’s disease patients and 27% of UC patients also experienced gluten sensitivity. In addition, consuming gluten was associated with recent flare-ups among the patients studied.
In this case study, a 28-year-old man was admitted to the hospital for a severe flare-up of UC. He was experiencing bloody diarrhea despite strict adherence to his prescribed drug protocol, which included a topical anti-inflammatory and an immune suppressant. A thorough examination revealed severe damage in his colon. Based on blood tests and a small intestine biopsy, celiac disease was ruled out. However, before moving toward more aggressive medical treatments, the young man decided to try a gluten-free diet. Within 6 weeks, he no longer had bloody diarrhea. Within 12 weeks, he was in full remission. When he started eating gluten again, his symptoms returned within 12 weeks. But he was able to induce remission with a gluten-free diet a second time.
While this is just one case, it’s a very interesting one to consider. Because this gentleman was on prescription medications for 4 years to manage his condition. And this is problematic for several reasons.
First, these drugs can cause nutrient deficiencies.
Second, suppressing your immune system for an extended period of time can lead to other serious problems.
And third, this case shows that these drugs were only masking his symptoms. They weren’t addressing the underlying source of inflammation. And in his case, it appears as if gluten was a contributing factor. Even in the absence of a classic celiac disease diagnosis.
Because gluten sensitivity symptoms are broad and diverse, non-steroid anti-inflammatory medications (NSAID) are often prescribed. And NSAIDS are associated with IBD.
This study found that about 40% of patients with IBD took NSAIDS regularly. It was also reported that taking NSAIDS more than 5 times a month increased the risk of a flare-up. Interestingly, acetaminophen (tylenol) use was also linked to active cases of IBD.
NSAIDS may help reduce inflammation, but they do so by blocking the production of prostaglandins. And prostaglandins stimulate the production of mucus in the digestive tract, which helps protect the lining from damage. This is why intestinal bleeding is a known side effect of long-term NSAID use.
Gastroesophageal Reflux Disease (GERD)
Gerd is most commonly described as persistent acid reflux. It’s diagnosed when acid and undigested food comes up from your stomach into your esophagus on a regular basis. And when this happens, it causes a burning sensation. Over time, the acid can damage the lining of your esophagus, which causes inflammation.
Many people believe GERD is caused by the production of too much stomach acid. But in most cases, it’s the opposite. Instead, it occurs because your food is not getting digested properly. So it ends up sitting in your stomach and eventually travels up your food pipe. This may happen because you lack stomach acid. Gluten-induced digestive dysfunction may also be a contributing factor.
In this study, 12% of celiac patients were also diagnosed with peptic disease, which includes GERD, heartburn, and stomach ulcers. And 79% with this group tested negative for H. pylori, a bacteria known for contributing to peptic diseases.
This is important information because when patients present with symptoms of peptic disease, doctors most often only test for H. pylori infections. But this study suggests that patients should also be screened for gluten sensitivity, especially if an H. pylori test is negative.
In this study, 30% of patients with celiac disease had moderate to severe GERD symptoms as compared to 5.7% of healthy controls. When the celiac patients with GERD were put on a gluten-free diet, their symptoms subsided within 3 months. A majority of the patients remained symptoms free over the course of 4 years without the need for proton pump inhibitor medications.
Another study found that a gluten-free diet significantly reduced the relapse rate of GERD among patients with celiac diease when compared to non-celiac patients (25% versus 71% respectively). Thus, the researchers concluded that celiac disease could be a risk factor for developing GERD.
It’s also worth noting that common medications prescribed for GERD have serious risks, especially when taken long term. For example, if you shut down stomach acid production, it makes it harder for your body to digest food. Antacids can also cause nutrient deficiencies, including calcium, vitamin B12, folate, iron, and zinc.
Esophagitis (Inflamed Esophagus)
Esophagitis is characterized by inflammation of the esophagus. It causes pain and difficulty swallowing. It can also cause food to get stuck in your esophagus.
The inflammation can be caused by an infection or acid reflux. And I just explained how reflux can be triggered by gluten.
In addition, this study found damage to the lining of the esophagus among celiac patients with and without reflux symptoms was more prevalent when compared to those without celiac disease. Also, the damage was repaired after the implementation of a gluten-free diet. This suggests that gluten-induced celiac disease can cause damage to other parts of the digestive tract, including the esophagus.
Eosinophilic esophagitis (EE) is also a condition that causes inflammation in the esophagus. However, it’s caused by a build-up of white blood cells typically due to an allergic reaction to a food or other environmental allergens. Thus, like gluten sensitivity, it’s an immune-related disease.
The prevalence of EE is higher (~10%) among those with celiac disease than the general population. Thus, researchers suggest that celiac patients should also be regularly screened for EE to prevent complications.
Interestingly, a study of children with both celiac disease and EE found that a gluten-free diet was just as effective as treatment with proton pump inhibitors or steroid medications. And this study reported similar results.
This study examined adults with EE and found the percentage of those with celiac disease to be higher than reported in the general population (8% versus 1%). And fifty percent of patients with both EE and CD successfully treated EE with a gluten-free diet. So in some cases, it may be that gluten or wheat acts as an allergen.
Diverticulitis
When regions of your digestive tract are damaged, the tissue can bulge out and form pockets known as diverticula. Diverticulitis occurs when the diverticula become inflamed and/or infected. It’s one of the most common gastrointestinal disorders in the Western world.
Diverticulitis is usually accompanied by pain, constipation, diarrhea, and bloating. Bloody stool is also a possible symptom.
So what causes the diverticula to form in the first place? Could it be gluten-induced gut inflammation that weakens the integrity of the digestive tract? Well, research has linked chronic gut inflammation with diverticulitis.
In this case study, a woman reported to the ER with abdominal pain, vomiting, and nausea. During the course of her visit, her pain increased as well as her white blood cell count, indicating a possible infection. She underwent emergency surgery. And the doctors found diverticula on her small intestine that had ruptured. So they removed the affected portion of her small intestine.
Twenty days later, the woman complained of abdominal pain, gas, and frequent diarrhea. Based on a biopsy and the presence of anti-gluten antibodies, she was then diagnosed with celiac disease. After two months on a gluten-free diet, her symptoms went away. And her vitamin B12 levels returned to normal.
The doctors suspect this patient had a silent case of celiac disease that was triggered by the surgery. And suggest that patients should be tested for celiac disease before undergoing gastrointestinal surgery to prevent complications. Nevertheless, it’s clear that this woman had a concurrent case of both diverticulitis and celiac disease.
In this case study, a young boy with abdominal pain and weight loss was also diagnosed with both diverticula on the small intestine and celiac disease. Due to a long history of malabsorption, the doctor suggested that celiac disease preceded the diverticula. And an improvement in symptoms was reported after a gluten-free diet was started.
Another potential cause of diverticulitis may be gluten-induced gut bacteria imbalances that can slow down your bowels, which can then causes waste to build-up in the diverticula and become inflamed.
Low fiber diets have long been associated with diverticulitis. The theory states that they can cause constipation and a build-up of waste in your colon as well as gut bacteria imbalances. Thus, high fiber diets with lots of whole grains are often prescribed to manage the disease and prevent flare-ups.
However, a more recent study found that high fiber diets may not be beneficial for those with diverticulitis, especially when the fiber comes from grains.
Hemorrhoids
Hemorrhoids are swollen veins around your anus and in your rectum. They can be itchy and painful. They can also make bowel movements difficult.
Hemorrhoids occur due to increased pressure in your rectum, which can be from straining during a bowel movement as well as chronic constipation or diarrhea. And for those with gluten sensitivity, these bowel irregularities are a common symptom that could contribute to the formation of hemorrhoids.
Can Gluten Cause Gut Inflammation?
Yes. Directly and indirectly.
The key pathways are inflammation, nutrient deficiencies, and gut bacteria imbalances. Beyond gluten, the chemical pesticide, glyphosate, commonly applied to gluten containing grains during growth and harvesting, may also contribute to IBD.
So rather than rely solely on prescription medications, it’s important to determine whether or not gluten could be the underlying cause of your problems. Because once you remove your triggers, your gut will be able to heal.
Did going gluten free help you overcome IBD? Post your experience in the comments below.





4 Responses
I’ve been gluten free for 10 years. I still have problems with constipation and hemorrhoids. I realized I needed to go low oxalate recently because of oxalate crystals in my urine causing neuropathy and neuroma. I believe I may have leaky gut. Our bodies are so complex. I cut out all grains and many other things this summer based on your No Grain No Pain book.
I’m stumped. Maybe it takes a while for the oxalate crystals to leave my system. I cut out high oxalate foods about 3 weeks ago.
Any suggestions?
What a amazingly informative article. It has given me lot to think about and hope that my conditions can be addressed.
I got hyperthyroidism and stomach inflammation
My test came back to be Celiac Sensitive. In this article, I have no constipation, and other symptoms for celiac.